 Assistant Professor
Assistant ProfessorPh.D. Albert Einstein College of Medicine
Basic Science Tower, T-5, Room 120
Stony Brook University
Stony Brook, NY, 11794-8661
Phone: (631) 444-6075
Maricedes.Acosta@stonybrook.edu
Department of Physiology & Biophysics
Research
Neuroendocrine regulation of the hypothalamus-pituitary-gonad (HPG) axis
Research in this laboratory investigates the interplay between metabolism and reproductive physiology. Although the mechanisms mediating the influence of metabolism and nutrition on fertility remain unclear, a strong association between metabolic disorders and infertility exists. For example, conditions of negative energy balance such as food restriction are associated with inhibition of gonadotropin release and hypogonadism. Similarly, obesity or excessive body fat can also negatively affect reproductive function in mammals; alterations in luteinizing hormone (LH) release are often observed. We hypothesize that many cases of infertility result from alterations in the regulation of gonadotropin releasing hormone (GnRH) secretion by peripheral metabolic signals. Currently, there is a high incidence of obesity and type 2 diabetes in our society. These diseases exacerbate endocrine disorders such as polycystic ovarian syndrome, which is associated with reduced fertility in women. Therefore, our research is highly relevant in attempting to delineate the pathways by which metabolic dysfunction may give rise to infertilities associated with altered GnRH secretion.
A major focus of this laboratory is to study the role of the enzyme phosphatidylinositol-3-kinase (PI3K) in the central control of the HPG axis. It is well known that hormones such as insulin, leptin and insulin-like growth factor-1 (IGF-1) act in the brain, especially the hypothalamus, to regulate the HPG axis. However, the intracellular signaling mechanisms by which their actions are coordinated are unknown. PI3K activity is shared by a diversity of signaling pathways including those initiated by insulin, leptin and IGF-1. Therefore, we hypothesized that PI3K is a key integrator of metabolic and neural signals regulating GnRH secretion. We use transgenic animal models to identify the neuronal populations in which activation of PI3K signaling regulates reproductive function. The kisspeptin and galanin-like peptide (GALP) neurons in the hypothalamus are strongly linked to the initiation of puberty and maintenance of fertility. By taking advantage of the Cre-Lox P system we are investigating the effect of neuron-specific deletions of PI3K catalytic subunits (p110α and p110β) on the reproductive axis. We also utilize imaging and molecular biology tools to monitor the activation of PI3K signaling in these neurons.
A second goal of our laboratory is to study how steroid hormones regulate intracellular signaling pathways such as PI3K. For example, in addition to its role in feedback control of gonadotropin release and reproduction, the steroid hormone estradiol-17 β (E2) has diverse physiological functions that include control of sexual differentiation during development, the regulation of mood, cognitive function, memory, behavior, appetite and metabolism. These E2effects are largely dependent on the activation of estrogen receptors (ERα and ERβ) in specific neuronal populations. The PI3K signaling pathway is suspected to play a central role in many of these E2 regulated physiological responses. Therefore, our studies are designed to investigate the links among E2, PI3K signaling, and the control of reproductive function and metabolism.
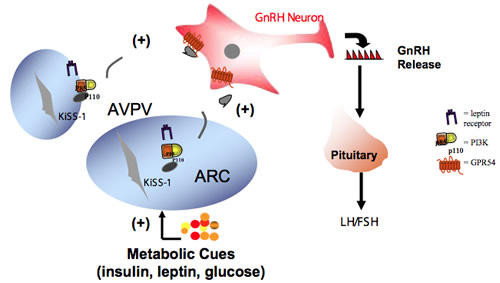
Fig. 1. Model proposing that GnRH neurons receive information from peripheral metabolic cues indirectly through the activation of PI3K signaling in kisspeptin neurons. Kisspeptin neurons reside in the AVPV (anteroventral periventricular nucleus) and ARC (arcuate nucleus of the hypothalamus), the latter containing functional leptin receptors.
- Publications
- Laboratory Personnel
-
- Acosta-Martínez M., Fiber J.M., Brown R.D. and Etgen A.M., (1999). “Localization of α1B – Adrenergic Receptor in Female Rat Brain Regions Involved in Stress and Neuroendocrine Function.” Neurochemistry International, Volume 35(5):383-392.
- Acosta-Martínez M., A.M., Etgen, (2002). “Activation of μ-Opioid Receptors Inhibits Lordosis Behavior in Estrogen and Progesterone-Primed Female Rats.” Hormones and Behavior, Volume 41(1):88-100.
- Acosta-Martínez M., A.M., Etgen, (2002). “The Role of δ-Opioid Receptors in Estrogen Facilitation of Lordosis Behavior.” Behavioral Brain Research, 136 (1): 93-102.
- Acosta-Martínez M., A.M., Etgen, (2002). “The Effect of Estrogen on μ-Opioid Receptor-Stimulated [35S]- GTPγS Binding in Female Rat Brain.” Neuroendocrinology, 76(4): 235-242.PHS 398/2590 (Rev. 11/07).
- Acosta, Maricedes 5. Etgen, A. M. and Acosta-Martínez M., (2003). “Participation of Growth Factor Signal Transduction Pathways in Estradiol Facilitation of Female Reproductive Behavior.” Endocrinology, 144(9):3828-3835.
- Acosta-Martínez, M., O. Gonzalez-Flores, and A.M., Etgen, (2006). “The role of progestin receptors and the mitogen-activated protein kinase pathway in δopioid receptor facilitation of female reproductive behaviors.” Hormones and Behavior, 49:458-462.
- Acosta-Martínez, M., Horton, T.H. and Levine J.E. (2007), Estrogen receptors in neuropeptide Y neurons: at the crossroads of feeding and reproduction. Trends Endocrinol Metab.Mar;18(2):48-50.
- Acosta-Martínez, M. and Levine, J.E. (2007), Regulation of mediobasal (MBH) KATP channel subunit gene expression by hyperglycemia in the hypothalamus of female rats. AJP,Endocrinology and Metabolism Jun;292(6):E1801-7.
- Huang, W., Acosta-Martínez, M., and Levine, J.E. (2008), Ovarian steroids stimulate KATP channel subunit gene expression and confer responsiveness of the GnRH pulse generator to KATP channel modulation. Endocrinology May;149(5):2423-32.
- Foecking, F. McDevitt, M.A., Acosta-Martínez, M., Horton, T.H. and Levine, J.E. (2008) Neuroendocrine consequences of androgen excess in female rodents. Horm. Behav.May;53(5):673-92.
- Huang, W., Acosta-Martínez, M., Horton, T.H. and Levine, J.E. (2008), Fasting-induced suppression of LH secretion does not require modulation of ATP-sensitive potassium (KATP) channels. Am J Physiol Endocrinol Metab 295:1439-1446.
- Acosta-Martínez, M., Luo J., Elias, C., Wolfe, A., and Levine, J.E. (2009), Male-biased effects of GnRH neuron-specific deletion of the PI3K regulatory subunit p85α on the reproductive axis.Endocrinology. Sep;150(9):4203-12.