Welcome to the Williams Translational Nanomedicine Lab.
Kidney-Targeting Nanomedicine
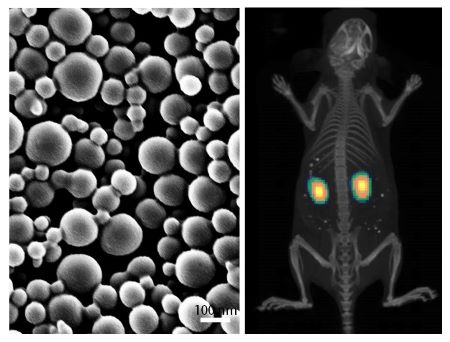
The Williams Lab designs and tests novel, safe, and biocompatible polymeric mesoscale nanoparticles that target the kidneys. Given that the overwhelming majority of nanoparticles localize to the liver and spleen, this kidney-targeting behavior is unique, enabling a wide range of therapeutic possibilities. As treatment options for acute kidney injury and chronic kidney disease are currently limited, the Williams Lab is exploring these nanoparticles as a platform for the treatment of such ailments. We are working with nephrologists and kidney disease biologists at Stony Brook and across the country to improve renal health.
We aim to develop targeted therapeutic versions of various biologic and small molecule targets to the site of acute and chronic renal diseases. This involves enhancing renal targeting through novel particle design and in vitro evaluation. Particle safety, pharmacology, and therapeutic efficacy are investigated in established rodent models of renal diseases. Ex vivo and in vivo imaging and endpoint evaluations complement and inform novel therapeutic targets and efficacy studies. We aim to bring these translational therapeutics to the clinic to improve patient outcomes.
Diagnostic Nanomedicine
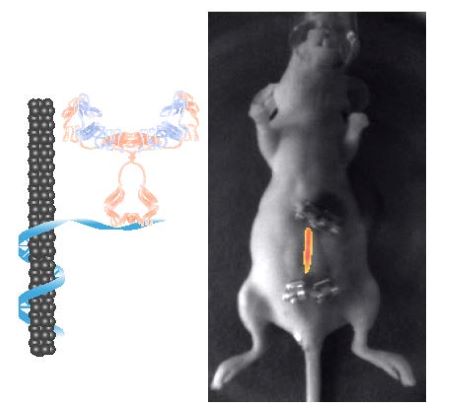
The Williams Lab engineers and optimizes rationally-designed, biocompatible nanosensor to improve diagnostics in cancer and other inflammatory diseases. We are working to design single-walled carbon nanotube (SWCNT) sensor platforms for on-board and bedside disease diagnostics. We also use these sensors as tools to understand disease biology and to study drug pharmacology. We are working with oncologists and disease biologists to increase early detection and improve therapeutic development.
We aim to develop near-infrared fluorescent nanosensor tools that change color or intensity in the presence of cancer biomarkers, cytokines, therapeutic drugs, or other disease-relevant analytes. Fluorescent SWCNT are optimized with molecular recognition elements such as antibodies or aptamers, screened to prevent non-specific adsorption, and tested in animal serum, human samples, and in mouse and rat models of disease. We aim to bring these diagnostic sensors to the clinic for cancer or chronic inflammatory disease, as tools to monitor therapeutic efficacy, and to other scientists to monitor the role of cytokines in disease.
CONTACT US:
Ryan M. Williams, Ph.D.
Office: (631) 444-9364
Email: Ryan.Williams@stonybrookmedicine.edu