Mission
To advance emergency care through innovative research that improves patient outcomes, enhances healthcare delivery, and shapes the future of acute care medicine.
Vision
The Center for Research and Clinical Trials is dedicated to conducting and supporting high-quality, inclusive, collaborative, and impactful research in acute care. We strive to generate – and to help others generate – evidence that informs clinical practice, drives innovation, promotes health equity, and prepares the next generation of clinician-scientists to improve emergency care for all patients.
Who We Are and What We Do
The Center for Research and Clinical Trials is a multidisciplinary team of physicians, nurses, scientists, coordinators, and support staff dedicated to advancing knowledge in acute care medicine. The division
designs, conducts, and supports clinical and translational research that addresses the urgent needs of patients presenting to the emergency department.
Our team specializes in implementing investigator-initiated studies, industry-sponsored clinical trials, federally-funded studies, and collaborative multicenter research. With expertise in trial design,
regulatory compliance, data management, and patient engagement, we ensure that research is conducted with the highest ethical and scientific standards.
In addition to fostering the generation of evidence that informs clinical practice, the center provides education and mentorship for trainees and early-career investigators, building capacity for the future of emergency medicine research.
Our department was among the first to develop an Academic Associates (AA) program in which undergraduate students get university credit in exchange for working shifts in the Emergency Department. While on duty, the AAs screen patients for eligibility to participate in any of the ongoing departmental research studies. While they primarily focus on non-funded investigator-initiated projects, they also assist the research nurses and coordinators in identifying patients that could be eligible for funded studies. For most observational studies, the AAs consent and collect data. autonomously. They
are also responsible for entering the data into electronic databases such as REDCap. The program has been established to introduce undergraduate students to clinical research while facilitating patient enrollment. The program offers academic credits and hands on experience.
Use of the Academic Associates for your project requires approval from the Research Steering Committee and the Director of the Academic Associates program. If approved, it is the responsibility of the Principal Investigator (PI) to in-service the academic associates and conduct oversight of their work.
Periodic study updates to the academic associates should also be the responsibility of the PI.
The Academic Associates program continues throughout the academic year, and the AAs are present in the ED Monday-Friday from 09:00-21:00.
The primary role of the research nurses is to facilitate conducting industry sponsored and federally funded research studies. Their responsibilities include assisting the principal investigators in preparing and submitting the IRB, coordinating site initiation visits, consenting patients, collecting data, administering study therapies (when appropriate), and completing data collection forms. They attend investigator meetings and conduct patient follow-up visits as per study protocol.
The role of the departmental statistician is to assist investigators in designing their study, defining outcomes, and choosing the appropriate tests to analyze their data. They will also assist in calculating the study sample size. After study completion, the statistician will assist in performing data analysis and
preparing tables and figures for abstract presentations and manuscripts.
The role of the administrative assistant is to assist faculty, fellows, and residents prepare and submit their IRB applications. The administrative assistant also assists investigators in developing study budgets and preparing and submitting research grants. The administrative assistant can also assist in
retrieving publications from the library.
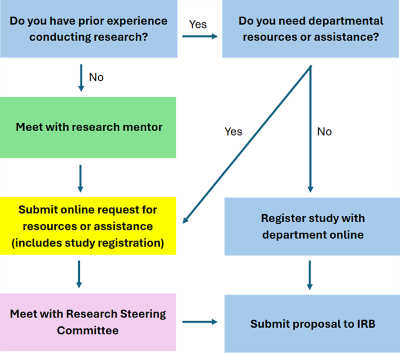
The role of the Research Steering Committee is to support and encourage faculty to design and conduct high quality research that aims to improve patient care. Committee members include senior and junior faculty with considerable experience designing and conducting research studies. They will meet with individual faculty members and assist them in successfully moving forward with their projects. At times, when departmental resources are limited or already heavily engaged the Committee will help prioritize the various projects with the goal of supporting as many projects and faculty as possible emphasizing those projects most likely to succeed and most likely to have an impact on our patients.
Committee members:
Imo P. Aisiku, MD, MSCR, MBA (System Chair)
Adam J Singer, MD (Committee Chair)
Samita Heslin, MD, MBA, MPH, MA, MS
Zhiyuan Zhang, MD, PhD
Lauren Maloney, MD
Erika Newton, MD, MPH
Brian McMahon, MD
Deborah LeBlanc, RN
Julian Hise, BS
Learn more about what you need to know to meet your resident scholarly activity requirement
Procedure For Submitting New Research Proposals